Last month the federal government announced that the AstraZeneca vaccine would be phased out, with it being available only ‘on request’ after October of this year. This decision followed numerous blood clotting cases and some deaths occurring following vaccination.
The government will now rely on the Pfizer and Moderna vaccines. However, these mRNA inoculations are not without their problems, either. Indeed, the US Food and Drug Administration has added a warning about the risk of developing heart inflammation to information about the Moderna and Pfizer vaccines.
The FDA announced in June that it would add the warning after the Centers for Disease Control and Prevention had reported that more cases of heart inflammation – either myocarditis or pericarditis – were found in young adults and children after they received the vaccines, which use mRNA technology.
The warning issued by the FDA says that there may be increased risks “particularly following the second dose and with [the] onset of symptoms within a few days after vaccination.”
There have been more than 1,200 cases of pericarditis or myocarditis in individuals who are aged 30 or younger who have received the vaccine doses, according to CDC findings released in the second half of June.
This comes on top of the revelation that more than 4,000 people have been hospitalized or died from COVID-19 “breakthrough” cases, according to the CDC.
In a late June update, the agency said that 4,115 people have been hospitalized or have died from COVID-19 despite having been fully vaccinated.
The total number of individuals who have died from COVID-19 after being vaccinated is 750, according to the CDC. It noted that 76 percent of hospitalizations and deaths from breakthrough cases occurred among individuals over the age of 65.
“As of June 21, 2021, more than 150 million people in the United States had been fully vaccinated against COVID-19,” said the agency in its June 25 update. “During the same time, CDC received reports from 47 U.S. states and territories of 4,115 patients with COVID-19 vaccine breakthrough infection who were hospitalized or died.”
Naturally, American health officials have trotted out the Mandy Rice-Davies response, downplaying the new data and saying that they had anticipated breakthrough cases, illnesses, and deaths.
“To be expected,” Dr. Paul Offit, a top advisor to the FDA on children’s vaccines, told CNBC on June 25. “The vaccines aren’t 100% effective, even against severe disease. Very small percentage of the 600,000 deaths.”
In fact, on May 1, the CDC announced that it had stopped counting all breakthrough cases and only reports on those that lead to hospitalization or death.
This revelation came around the same time Israeli officials said that during a recent outbreak of COVID-19, about half of the adults infected in the recent outbreak were fully vaccinated.
A report quoting Ran Balicer, who leads an expert advisory panel for the Israeli government, in announcing the finding on June 25, said that the country might end up in lockdown again. Balicer and other officials said that 90 percent of the new cases were caused by the so-called Delta COVID-19 variant. Israel, seen as the “gold standard” in rolling out its vaccination programme – more than 70% of the population has had the jab – relied heavily on both the Pfizer and Moderna jabs.
Of course, officials are dismissing most of these “breakthrough” infections, since they are asymptomatic and nothing to worry about. How the narrative has changed. Up until now, numbers of positive cases, even if most of them were asymptomatic, were seen as justification for panic lockdowns and other coercive measures.
The million dollar question is, if these vaccines are ‘safe and effective’ – 95% according to some, why are serious illness and deaths occurring from the very infection these vaccines are supposed to protect against?
The answer was alluded to by Ramesh Thakur in these pages last month. The trick, as it were, lies in the rubbery figures behind the ‘relative reduction rate’ versus the ‘absolute reduction rate’. When one analyses the New England Journal of Medicine report which published the Pfizer trial numbers, this trick becomes clearer, as explained to me by an immunologist friend who, for professional reasons, wishes to remain anonymous.
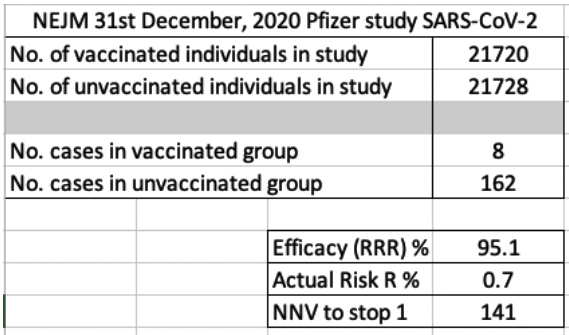
My friend created an Excel spreadsheet calculating the RRR (Relative Risk Reduction – Efficacy %) and the ARR (Absolute Risk Reduction %). The first screen shot below shows these two values based on the data published. The NNV (number needed to vaccinate to stop 1 case) is 141 individuals.
This corroborates the finding of a Lancet study referred to by Ramesh Thakur in last week’s magazine. The study calculates that for one additional person to be protected against Covid, around 100 will have to be vaccinated.
Further, the immunologist asserts that the definition of a ‘case’ is very rubbery. The classical herd immunity definition of a case is anyone who is symptomatic and can therefore transmit the illness to a non-immune individual. This definition doesn’t account for the possibility of asymptomatic spread and the vaccine trials also haven’t assessed for this possibility.
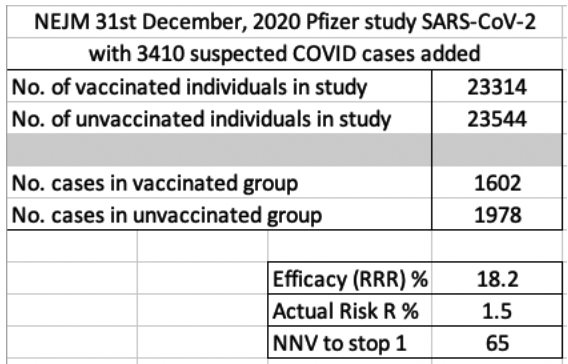
Interestingly, he advised about one other piece of information that is not in the NEJM article, but was submitted to the FDA for emergency use authorisation. This concerned the occurrence of 3410 suspected COVID cases amongst the trial participants. It turns out that none of these were tested to determine if their flu like illnesses were actually due to SARS-CoV-2. Instead all were excluded from the study and the calculations. There were 1594 in the vaccine group and 1816 in the placebo group. These numbers are included in the second screenshot below. Even using RRR, the results are woeful (efficacy of 18.2%). The absolute risk reduction moves up a bit from 0.7% to 1.5%, but, in my friend’s view, it is still ridiculously insignificant.
Critically, as the NEJM report itself admits, “[T]his trial and its preliminary report have several limitations”. First among these that the group of participants in the trial is not large enough to detect less common adverse events reliably. To quote the report: “Therefore, both the occurrence of adverse events more than 2 to 3.5 months after the second dose and more comprehensive information on the duration of protection remain to be determined.”
In other words, there has been a distinct lack of time for proper evaluation of the adverse effects of these mRNA vaccines. As the reports cited above show, those effects, many of them fatal, are being seen now.
Dr Rocco Loiacono is a senior lecturer at Curtin University Law School.